Hyperspectral Imaging (HSI) is the process by which many (i.e., >50) narrow (e.g., 2-10 nm) samples of the electromagnetic spectrum are measured. So many samples make a representative spectrum. These data can be either collected over reflected (400-3,300 nm) or emitted (>3,300 nm – 15,000 nm) light, or both. The result of collecting all these individual spectra is a cube which can be visualized as in Figure 1.

Figure 1
Reflected light is light that originates externally and shines on a material’s surface. This light does one of three things: it’s absorbed, transmitted, or reflected. Since most light penetration is restricted to a few microns, reflected light contains information about the interactions of incoming photons with the surface material. Longer thermal infrared wavelengths (> 3,300 nm) are dominated by emitted light. These are thermal photons emitted by materials with a temperature above absolute zero.
The wavelength of the light dictates the resolution capabilities of the measured light. Shorter wavelengths (< 1000 nm) called Electronic Effects. These photons interact with an atom’s electron cloud. Absorbance features produced by this interaction are strongly dominated by intervalence charge transfer (IVCT).
There are a number of diagnostic absorption features between 2,000-2,500 nm. Many of these are combinations and overtones from primary absorption features at wavelengths > 3,300 nm. Thermal wavelengths (> 3,300 nm) measure molecular bond properties, i.e., bending, twisting, and stretching of molecules.
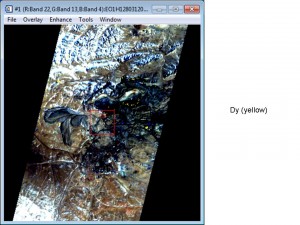
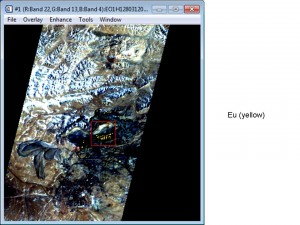
Below are examples of using Hyperspectral Imagery to find Rare Earth Elements.
All images can be clicked on to enlarge.
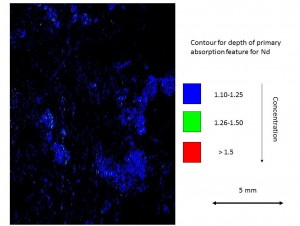
Microscope (click on photograph to enlarge)
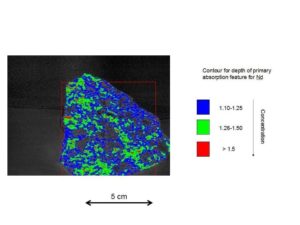
Handsample (click on photograph to enlarge)
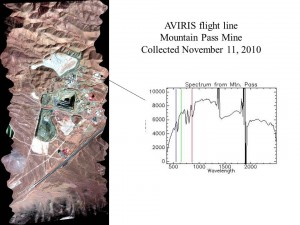
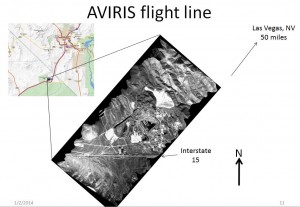
Airborne – AVIRIS (click on photograph to enlarge)
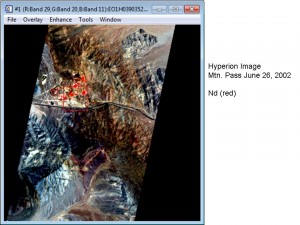
Satellite – Hyperion Imagery
Mountain Pass, California (click on photograph to enlarge)
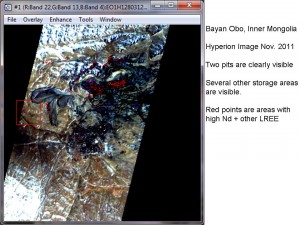
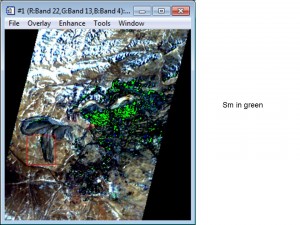
Bayan Obo Mine, China (click on photograph to enlarge)